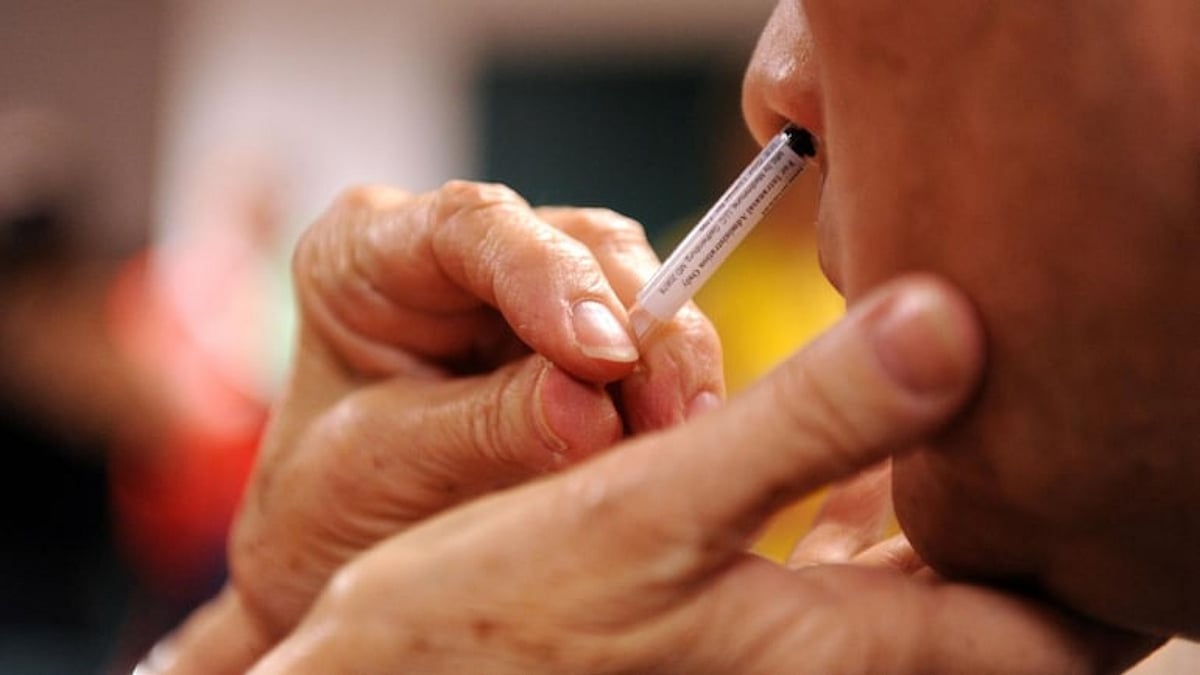
The first nasal vaccine against coronavirus developed by Bharat Biotech supported by Department of Biotechnology (DBT) and its PSU, Biotechnology Industry Research Assistance Council (BIRAC) has been given regulator's nod for conducting phase 2/3 trials, the Ministry of Science & Technology informed on Friday.
“This is the first of its kind COVID-19 jab to undergo human clinical trials in India. BBV154 is an intranasal replication-deficient chimpanzee adenovirus SARS-CoV-2 vectored vaccine. The Company has in-licensed technology from Washington University in St Louis, USA,” said the release.
The Phase 1 trial in healthy volunteers of age groups ranging from 18 to 60 years was well tolerated, according to the ministry of science and technology. The release said the doses of the vaccine administered to healthy volunteers in the Phase I clinical trial were well-tolerated. “No serious adverse events reported. Previously, the vaccine was found to be safe, immunogenic and well tolerated in the pre-clinical toxicity studies. The vaccine was able to elicit high level of neutralizing antibodies in animal studies," it said.
This is the first of its kind COVID-19 jab to undergo human clinical trials in India. BBV154 is an intranasal replication-deficient chimpanzee adenovirus SARS-CoV-2 vectored vaccine.
Bharat Biotech's Covaxin had demonstrated 77.8% efficacy against symptomatic Covid-19 and 93.4% against severe illness during the Phase 3 clinical trials.