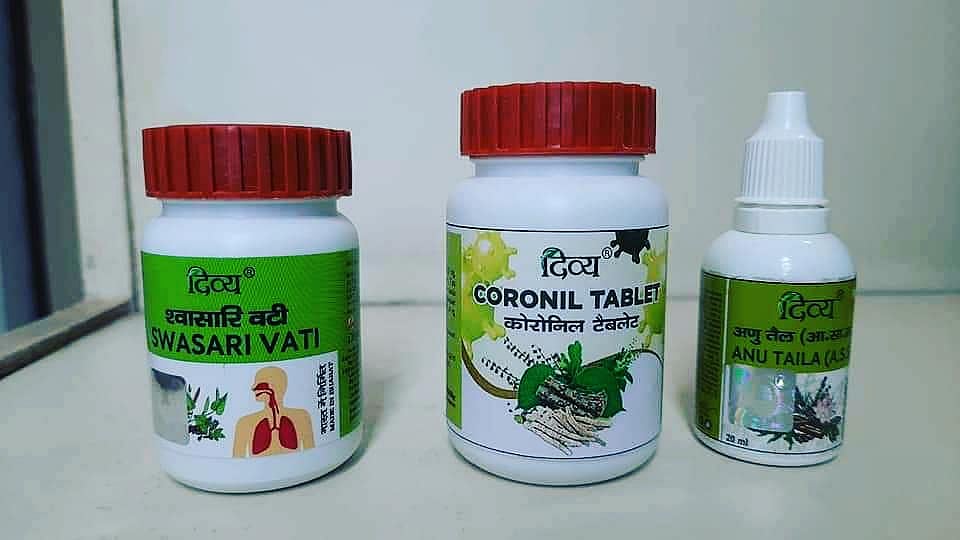
Ramadev Baba-led Patanjali responded to the notice issued by the Uttarakhand Drug department, stating that the company never promoted Coronil as COVID-19 medicines. The company even went on to state that the three medicines — Divya Swasari Vati, Divya Coronil Tablet and Divya Anu Tail were packed for convenience and were not promoted as medicine to fight COVID-19.
According to Zee report, the company told authorities that it only shared the benefits of the medicine and its successful test on COVID-19 positive people and never said that it can cure coronavirus. The company said this in a response to a notice which was issued by the Uttarakhand Ayush Ministry, soon after the launch of Patanjali’s Coronil. The authorities issued notice to Patanjali saying that the licence issued was not to find a cure for Coronavirus but to produce immunity booster kits and fever medicines.
“We never told the medicine (Coronil) can cure or control Corona, we said that we had made medicines and used them in clinical controlled trials which cured Corona patients. There is no confusion in it,” said Acharya Balkrishna, CEO, Patanjali.
The company had taken licenses for Divya Swasari Vati, Divya Coronil Tablet and Divya Anu Tai as a medication for common cold and cough. But went on to promote it in a way that led people to believe that it was used to treat COVID-19 patients.
The company claimed to have treated around 250 COVID-19 patients with these medicines and none of them lost their lives.
At present, the medication is not available commercially. During the launch, it was stated that it will be sold in Patanjali outlets soon and will be available for online purchase.