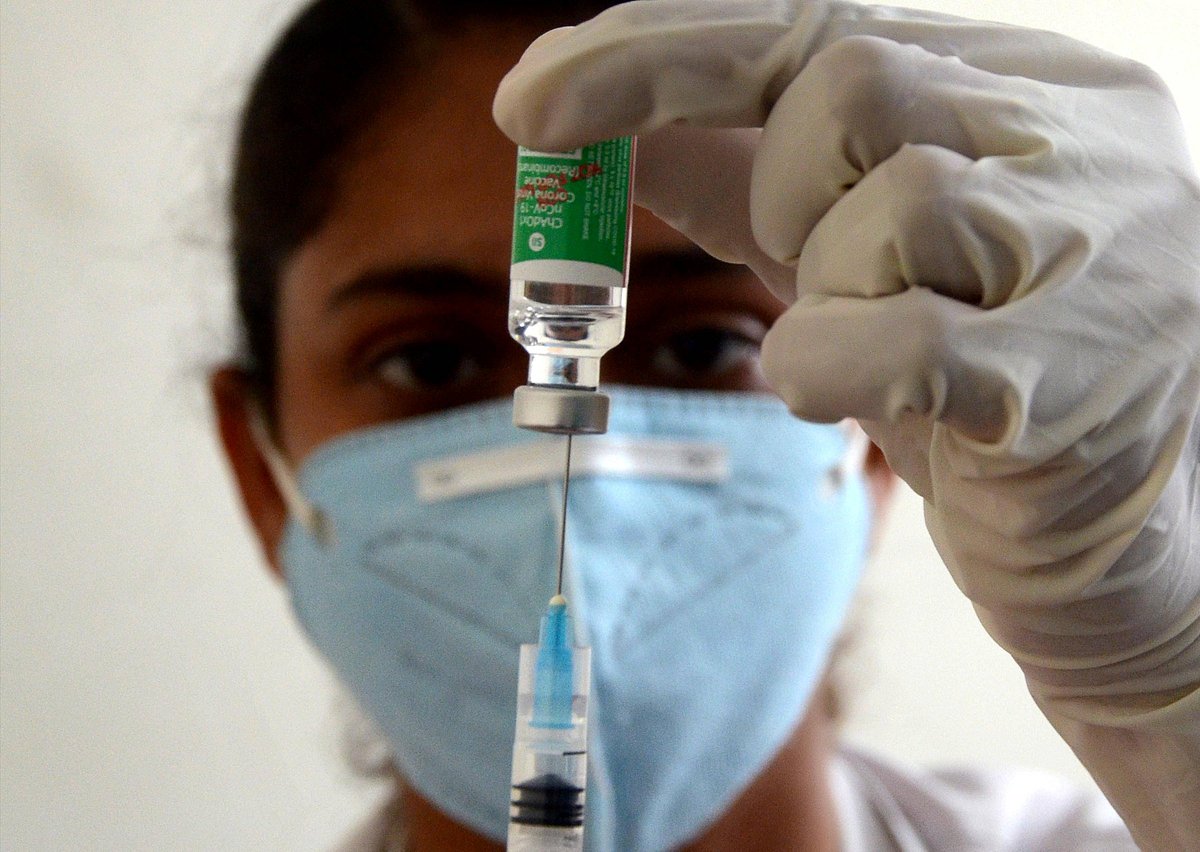
New Delhi: The Centre, in a counter-affidavit filed in the Kerala High Court on Friday, stated that the duration of 84 days between first and second dose of Covishield vaccine will provide the best protection against COVID-19, as per a Zeenews report.
The affidavit comes as a reply to a writ petition by Kitex Garments Ltd, Kochi, in which the company urged the government to allow it to administer the second dose of Covishield vaccine to its workers before the completion of the 84-day gap.
"India's National COVID Vaccination Program was built on scientific and epidemiological evidence, World Health Organization (WHO) guidelines and global best practices," Centre stated in the affidavit.
The affidavit also added that under the guidance of the National Expert Group on Vaccine Administration for COVID-19 (NEGVAC), the dose interval between the two doses of the Covishield vaccine had undergone a series of revisions based on the available and emerging scientific evidence.
"Based on the recommendations by NEGVAC, the schedule of the Covishield vaccine is to administer the second dose at 12-16 weeks interval after administration of the first dose. This is based on the technical opinion that the duration of 84 days between 1st and 2nd doses of Covishield is providing the best protection against COVID-19," the affidavit stated.
"To provide full vaccination coverage and facilitate international travel for genuine reasons, it was decided to allow the second dose before the prescribed time period of 12-16 weeks. As per the evidence available, the immunity provided by two doses of the Covishield vaccine with intervals less than 12-16 weeks would be better than partial vaccination. The decision to increase the Dose interval of the Covishield vaccine was based on scientific evidence and expert opinion as per available efficacy data," it further stated.
"No fundamental or statutory rights of the petitioner were infringed by the same. The grounds advanced by the petitioner in the writ petition were false and frivolous, hence deserves to be dismissed. The petition was totally misconceived/devoid of merits and deserved to be dismissed in the interest of justice," the affidavit added.
In the Kerala HC, the counsel for the petitioner submitted that the stipulation of the 84 day gap could not be made applicable to paid vaccines. The Counsel also pointed out that the medical protocol of the Covishield manufacturer had allowed a 28-day gap between the first and second doses of the vaccine.
According to media reports, the government was considering narrowing the gap between two Covishield doses for those 45 years or older on increased availability of the vaccine and mounting criticism that the longer gap was stipulated to overcome supply constraints without much scientific evidence backing it.
The government had drawn criticism for increasing the dosage interval as there was a shortage of vaccines in the country and the decision was seen as a move to gain time to procure more doses.
With ANI inputs





.png)



