Amidst all the hue and cry over the vaccine for COVID-19, the Nagpur bench of the Bombay High Court has asked Patanjali to follow the due procedure of law if it seeks to approve its medicine as a vaccine for the virus.
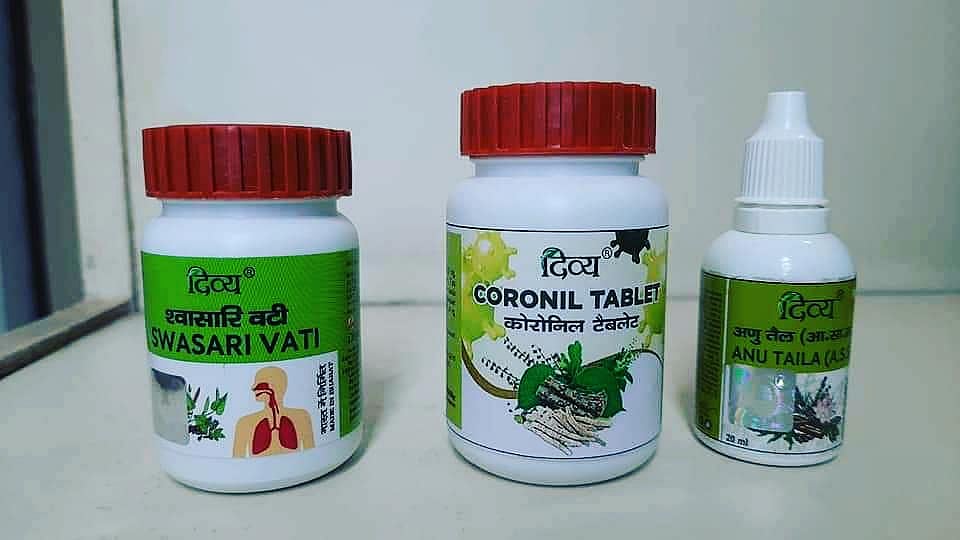
A bench of Justices Sanjay Gangapurwala and Rajendra Avachat was dealing with a PIL filed by a woman, seeking directives to the union government and other relevant authorities to start trial of Coronil to check its efficacy at the earliest. The PIL emphasised that, under these extraordinary circumstances caused by the pandemic, it is necessary to find a cure for COVID-19 at the earliest. "It is important to conduct research of the coronavirus medical kit / Coronil / Swasari Ayurvedic Product of Patanjali, to check its efficacy at the earliest," the PIL stated.
Accordingly, the woman told the judges that it is for the people to choose and decide which medical treatment to prefer - homeopathic, ayurvedic, allopathic and so on. "The authorities, thus, must be directed to conduct an appropriate scientific test immediately of the coronavirus medical kit / Coronil / Swasari," the lady urged.
At this, additional solicitor general SB Deshpande, appearing for the union government, told the judges that for a medicine to be approved, a long drawn process is prescribed. "The same cannot be completed in haste. Many trials have to take place," Deshpande said.
"The government has directed Patanjali not to promote the coronavirus medical kit / Coronil / Swasari as a medicine for COVID-19, but may use it as an immunity booster," the ASG submitted further.
Accepting his statement, the judges said, "If Patanjali has come out with some medicine, as it may claim, it has to follow all the procedure and the protocol for the approval of its medicine."
"The trials cannot be directed to expedite the protocol. The authorities will have to follow the protocol while approving a particular medicine," the judges added while noting that certain steps have already been taken with respect to the medicine as claimed by Patanjali and that certain directions are also issued.
"If the petitioner has some evidence of the efficacy of the medicine as contended, it may approach the authorities concerned," the bench said while disposing of the PIL.