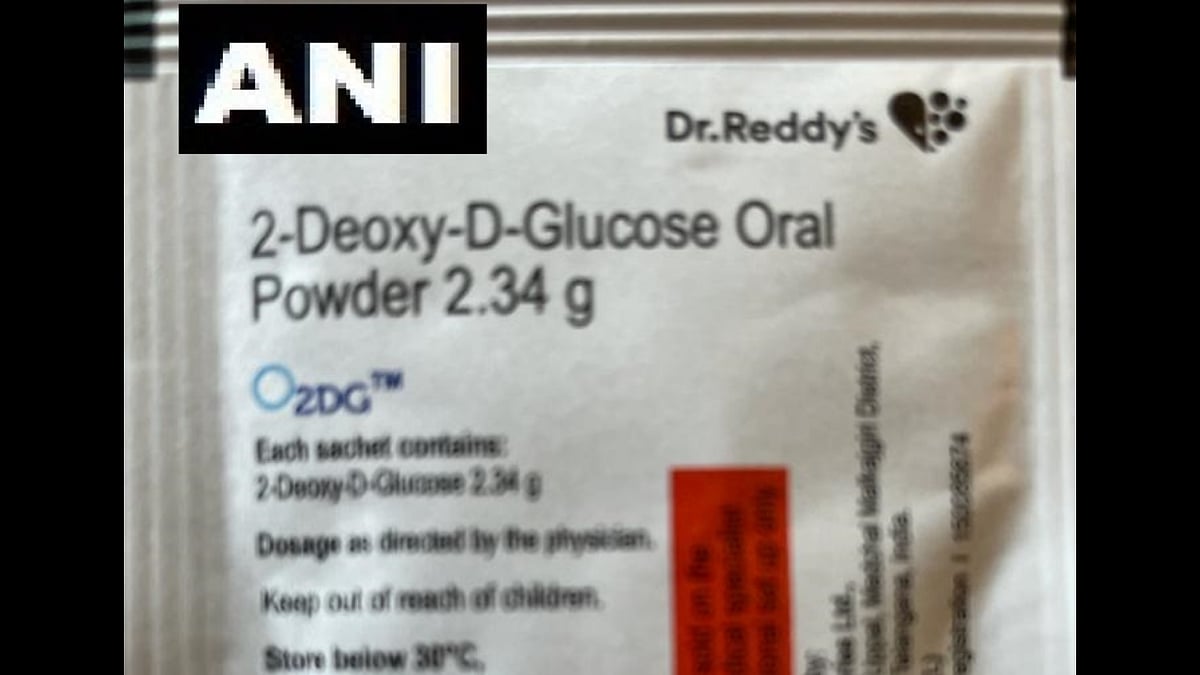
The Drugs Controller General of India that approved oral drug 2-deoxy-D-glucose (2-DG) developed by the DRDO for emergency usage to treat the COVID-19 infection was released by Defence Minister Rajnath Singh and Union Health Minister Dr Harsh Vardhan on May 17. The drug has been developed by DRDO's Institute of Nuclear Medicine and Allied Sciences (INMAS) in collaboration with Dr Reddy's Laboratories.
So far there had been no clear instruction given on the prescription of the drug. But no worries, the DRDO today released a list of guidelines that shows how the drug can be used by the COVID-19 patients, here'a look at it:
1. The 2DG is approved for Emergency use as adjunct therapy to the standard of care in the treatment of COVID patients in the hospital settings.
2. Ideally, 2DG should be prescribed as early as possible by Doctors for moderate to severe COVID patients for maximum duration up to 10 days.
3. Uncontrolled diabetes, severe cardiac problem, ARDS, severe hepatic and renal impairment patients have not been studied yet with 2DG, hence caution should be exercised.
4. 2DG should not be given to pregnant and lactating women and patients below 18 years.
5. Patients/Attendants are advised to request their hospital to contact Dr Reddy's Lab, Hyderabad for medicine supply at email: 2DG@drreddys.com
As per Dr Reddy's, the 'commercial launch' and supply of the drug to major government hospitals is expected to commence from mid-June. While, the price of DRDO’s 2DG anti-COVID 19 drug has been fixed at Rs 990 per sachet. The government officials said the central and state government hospitals would be provided with the medicine at a discounted price.
DRDO Chairman Dr G Satheesh Reddy had earlier told that DRDO and Dr Reddy's lab had gone through the complete trials and conducted trials across 30 hospitals and on a large number of patients.
The drug comes in powder form in a sachet, which is taken orally by dissolving it in water. It accumulates in the virus-infected cells and prevents virus growth by stopping viral synthesis and energy production. Its selective accumulation in virally infected cells makes this drug unique.